Safed, Israel -
One page proposal for support of a
Clinical trial for the prevention of respiratory failure and the need for ventilation in patients with Covid-19 infection.
Office of the Dean
The Azrieli Faculty of Medicine
Bar-Ilan University
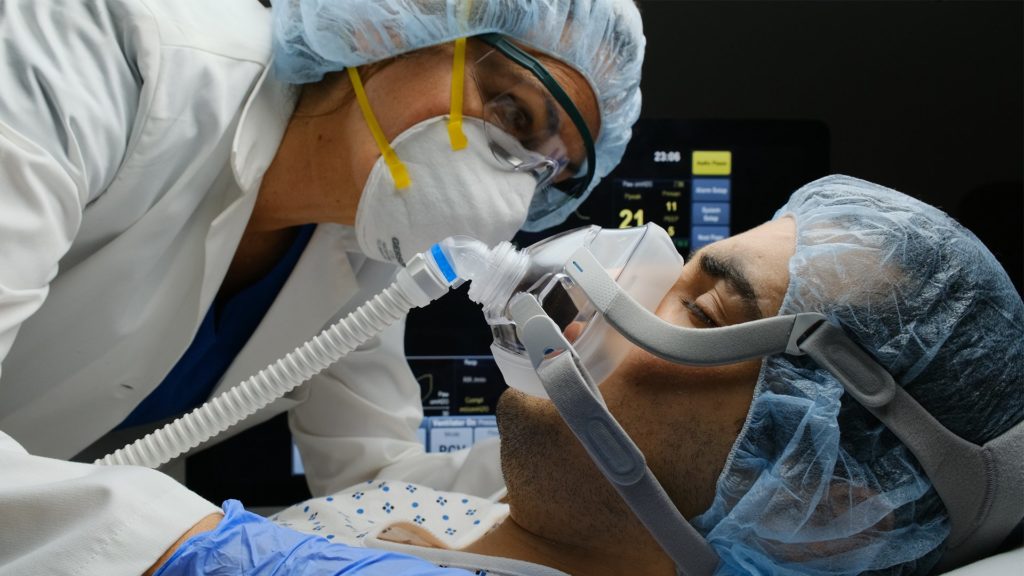
While many patients with Covid19 recover after a mild or moderate illness, a significant subset of patients progress to severe lung injury (Pneumonitis) with reduced ability to oxygenate, requiring connection to artificial respiration and ventilation, and in the most severe cases leading to death as a result of respiratory failure. A clinical research team at Bar-Ilan University’s Azrieli Faculty of Medicine led by Karl Skorecki (Dean of the Faculty of Medicine), in collaboration with colleagues at affiliated medical centers throughout northern Israel, have proposed and obtained Ministry of Health approval for a clinical trial to mitigate lung injury and prevent or lower the need for artificial respiration and ventilator support.
This proposal is based upon a protocol using a small peptide (Angiotensin 1-7), which naturally occurs in the body to protect the integrity of fragile lung cells and to maintain adequate blood flow to vital organs in situations of inflammatory over activity and blood vessel constriction. Based on examination of safety, the peptide has also already been approved by the FDA in the United States for use in clinical trials of a variety of lung and inflammatory states. However, never has there been so compelling and urgent a need.
The idea for this peptide’s lifesaving use in Covid-19 infection is based on the Bar-Ilan team’s longstanding knowledge and expertise with the physiologic pathway which generates Angiotensin 1-7, and which is nefariously utilized by the Corona virus. Specifically, the Corona virus enters lung cells by hijacking a particular membrane-bound enzyme (ACE2), which then carries the virus into cells where it propagates and generates a vicious feed-forward cycle. The more virus that is produced, the less the ACE2 enzyme can function, which results in depletion of the Angiotensin 1-7 peptide. As mentioned, it is this peptide which is powerfully anti-inflammatory, restores blood flow and is protective to lung cells – and so crucial for their integrity.
We believe that bypassing the disabled ACE2 and administering the Angiotensin 1-7 peptide by safe injection to the COVID-19 patient in need will support lung function until the patient is able to clear the virus from their body. Successful usage of this drug would reduce the need for ventilatory support and save lives.
On October 15, 2020, after careful inspection and review at the highest level of scrutiny for safety and benefit, the Israeli Ministry of Health issued its approval for the Bar-Ilan led team to proceed with a clinical trial. The trial protocol is simple: enroll 120 clinically eligible patients each for up to seven days, and measure outcomes. The protocol does not interfere with other ongoing treatment. It is anticipated that the clinical trial would take six to nine months to complete.
Your support and contribution to this clinical trial could potentially set the stage for a lifesaving intervention in this dreaded Covid-19 pandemic. Furthermore, and of crucial import, proof of success of therapy in this clinical trial would also be useful for any other future Corona based disease (as occurred for SARS in 2003, and is now recurring for Covid-19) since the treatment outsmarts the virus and bypasses the disabled ACE2 pathway.
Budget (USD) for the trial:
Clinical trial coordinators: 80,000
Angiotensin 1-7 production and delivery: 150,000
Biomarker monitoring and assays: 250,000
Data Analysis: 20,000
Total : 500,000
Bar-Ilan University (RA), The Azrieli Faculty of Medicine, Henrietta Szold st. 8, POB 1589 – Safed, Israel
Tel: 072 264 4937 • Dean.Medicine@biu.ac.il


